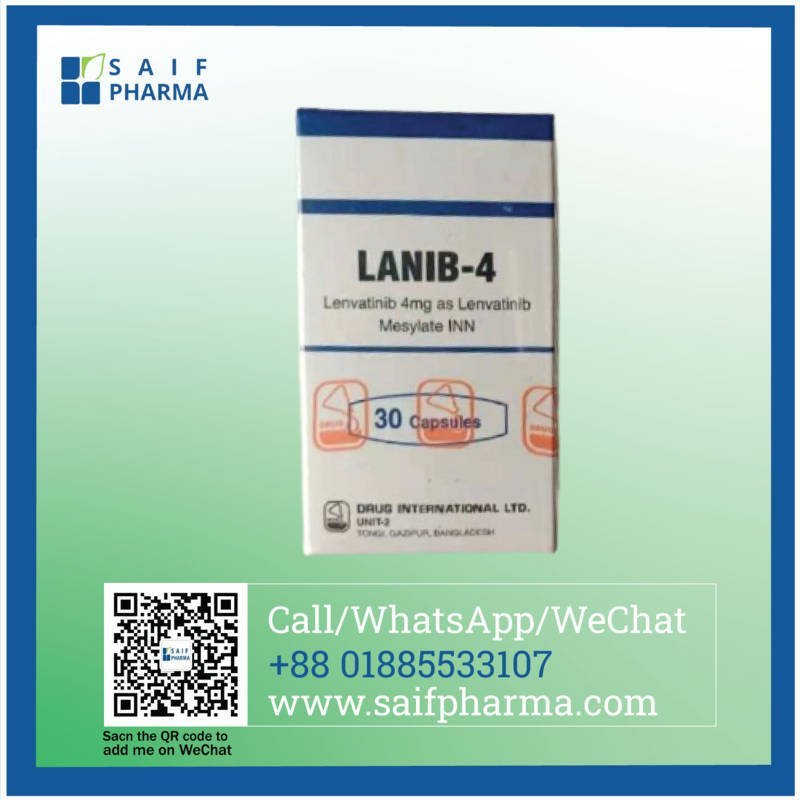
Each capsule contains Lenvatinib 4 mg as Lenvatinib Mesylate INN.
Lozenge & Administration
Recommended Lozenge for Discerned Thyroid Cancer (DTC) The recommended lozenge of Lenvatinib is 24 mg orally formerly daily until complaint progression or until inferior toxin. Recommended Lozenge for Renal Cell Melanoma (RCC) The recommended lozenge of Lenvatinib is 18 mg in combination with 5 mg Everolimus orally formerly daily until complaint progression or until inferior toxin. Recommended Lozenge for Hepatocellular Carcinoma (HCC) The recommended lozenge of Lenvatinib is grounded on factual body weight. Or, as directed by the registered croaker.
Suggestions
Discerned Thyroid Cancer Lenvatinib is indicated for the treatment of cases with locally intermittent or metastatic, progressive, radioactive iodine-refractory discerned thyroid cancer (DTC). Renal Cell Carcinoma Lenvatinib is indicated in combination with Everolimus for the treatment of cases with advanced renal cell melanoma (RCC) following one previous anti-angiogenic remedy. Hepatocellular Carcinoma Lenvatinib is indicated for the first-line treatment of cases with unresectable hepatocellular melanoma (HCC). Endometrial Melanoma Lenvatinib, in combination with Pembrolizumab, is indicated for the treatment of cases with advanced endometrial melanoma.
Use in Gestation & Lactation
Grounded on the medium of action, Lenvatinib can beget embryo-fetal detriment when administered to a pregnant lady. Pregnant women should be advised of the implicit threat to a fetus. Ladies of reproductive eventuality should be advised to use effective contraception during treatment with Lenvatinib and for at least 30 days after the last cure.